Content Attributes
Chemical species that donate or take electrons to establish a new chemical bond are electrophiles and nucleophiles. An electrophile is a molecule, ion, or atom that is lacking in electrons in some way. A nucleophile is a chemical species that donates an electron pair to establish a chemical bond in response to a stimulus.
A nucleophile is usually negatively or neutrally charged, with only a few donable electrons. Examples include H2O, -OMe, and -OtBu. The electron-rich is a nucleophile in general. Electrophiles are known for their positively charged, or they can also be neutral organisms with empty orbitals attracted to a Tron-rich circle.
Electrophile and nucleophile are terms used to describe chemical reactions that occur between electron donors and acceptors. These are the essential organic chemistry principles. They superseded the names cationic and anionic and were first used in 1933.
Learn how you can distinguish between Electrophile and Nucleophile components.
Electrophile
A chemical species that can tackle an electron pair that allows them to form bonds with nucleophiles is widely known as an electrophile. They are known as electrophiles as they receive electrons that are electrophiles Lewis acids. The majority of electrophiles are charged positively and consist of partial positive charges on an atom. However, it can even lack an octet of electrons.
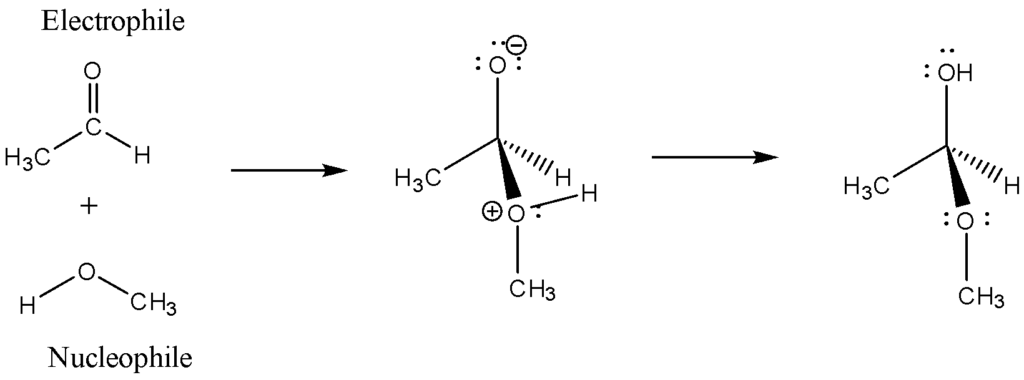
The most general interactions between the two (electrophiles and nucleophiles) are the addition and substitution procedure. Cations like H and NO usually polarise neutral molecules like HCl, acyl halides, carbonyl compounds, alkyl halides, and polarisable neutral molecules. Polarisable neutral molecules consist of Cl2 and Br2 and some oxidising agents, such as organic peracids and chemical species. These chemicals dose not persuade the octet rule in carbenes and radicals, whereas some Lewis acids like BH3 and DIBAL are all common electrophiles in organic.
- They have a lack of electrons and are drawn to them.
- They can be charged in either a positive or negative manner.
- Many electrons are attracted to atoms with many electrons, such as double bonds made of carbon and carbon, which have many electrons.
- The density impacts electron mobility, which occurs when electrons travel from a high-density to a low-density location.
- The use of electrophilic addition as well as the replacement processes should highly be encouraged.
- Lewis acids are named by the fact that they take electrons.
- Electrophiles are engaged in electrophilic substitution along with the addition procedure. They have a lack of electrons and are drawn to them.
- They can be charged in either a positive or negative manner.
- Electrons attack atoms with many electrons, such as carbon-carbon double bonds, which have a lot of electrons.
- The density impacts electron mobility, which occurs when electrons travel from a high-density to a low-density location.
- The use of electrophilic addition and replacement processes should be encouraged.
- Lewis acids are named by the fact that they take electrons
- Electrophiles are engaged in electrophilic substitution and addition processes.
Nucleophile
As previously stated, nucleophiles are electron-rich organisms with the ability to contribute electron pairs. All nucleophiles are Lewis Bases because of their electron pair donating tendency.
The word “nucleophile” is made up of two parts: nucleus and Philos. Philos is a Greek word that means “love.” As a result, nucleophiles are known as Nucleus Loving species. These nucleophiles might have a positive or negative charge.
- The affinity of a species for the positively charged nucleus is described by its nucleophilic nature.
- Nucleophilicity is a term that is used to compare the nucleophilic characteristics of various nucleophiles. It’s also known as a species’s nucleophile strength.
- Nucleophilic substitution is a process in which an electron-rich nucleophile selectively assaults a positively charged (or partially positively charged) atom in a molecule and bonds with the positively charged species to replace a leaving group.
- Ambident nucleophiles can carry out nucleophilic assaults from two or more separate locations in the molecule (or ion). These types of nucleophile attacks frequently result in the creation of several products.
Example:
Chlorine (Cl) has 3 lone pairs of electrons in just its atomic form. So it can donate them to other electron-deficient atoms or molecules by getting attached to them.
Factors of Differentiation Between Electrophiles and Nucleophiles
The factors to distinguish between Electrophile and Nucleophile is listed below:
| ELECTROPHILE | NUCLEOPHILE |
| Lewis acid is another name for it. | Lewis base is another name for it. |
| They have a positive/neutral charge. | They have a negative/neutral charge. |
| Electrophilic addition and electrophilic substitution processes are carried out on them | They are subjected to nucleophilic addition and substitution processes. |
| All carbocations | All carbanions |
| To establish a covalent bond, it accepts a pair of electrons. | It donates a pair of electrons to form a covalent bond |
| Electron-deficient | Electron-rich |
| Example: Hydronium Ion | Example: Chloride Ion |
The thiocyanate ion, with the chemical formula SCN–, is an example of an ambident nucleophile. This ion can target either the sulfur or nitrogen atoms with nucleophilic assaults. Creating a mixture of alkyl isothiocyanates with the chemical formula R-NCS and alkyl thiocyanates with the chemical formula R-SCN is common in nucleophilic substitution reactions alkyl halides involving this ion.
There were a few key distinctions between nucleophiles and electrophiles. When a ligand is activated by proximity to metal, it can be attacked promptly by a nucleophile or an electrophile. Each of these assaults can be a reaction to an abstraction or addition.
Nucleophilic & Electrophilic Substitution Reaction
On occasion, a compound is already joined to the positive area of another complex. A third negatively charged compound or lone pairs of electrons assaults the main molecule and replaces the negatively charged component that was previously there. The process is known as nucleophilic substitution.
For example, the positive area of CH3BR already has BR linked to it. The BR atom is replaced by the CN- atom when this chemical interacts with CN-. This leads to the chemical now becoming CH3CN.
The electrophile substitutes another electrophile already bonded to a molecule in the electrophilic substitution process. An electrophile, for example, can take the place of the H atom on a Benzene chain.
Conclusion
In chemical reactions involving different atoms or chemical species, electrophiles and nucleophiles play a significant role. Opposites, as we all know, attract. Electrophiles and nucleophiles are opposed. This attraction is what causes so many chemical reactions and complex formations.